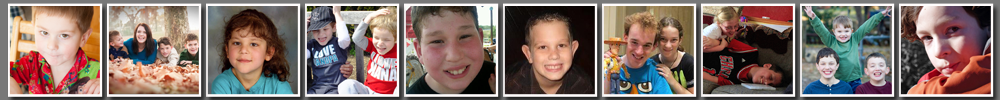
An Open Letter Regarding STX209 to Dr. Severin Schwan, Roche Pharmaceuticals
To see Dr. Schwan’s response to this letter please see here: Roche Response
Dr. Severin Schwan
Chief Executive Officer
Roche Pharmaceuticals
F. Hoffmann-La Roche AG
Konzern-Hauptsitz
Grenzacherstrasse 124
CH-4070 Basel
Schweiz
June 10, 2013
Dear Dr. Schwan:
Behind every drug Roche Pharmaceuticals makes are the personal stories of patients and their families. You must hear these stories everyday, and we cannot imagine the pressures you face determining where to put your resources and whose lives – whose children’s lives – you will try to save. We understand you cannot support every drug and every trial, but you are a father, so surely you will understand our desperation and need to reach out to you directly to help our children. Like most parents, we are certain you would go to any lengths – contact any person – if your children needed help; so you’ll understand why we are writing you this letter.
Recently, Roche Pharmaceuticals was involved with a Cambridge, Massachusetts-based pharmaceutical company called Seaside Therapeutics, Inc., which has been developing a drug called STX209 (Arbaclofen). This compound has been in Phase III trials for children with fragile X syndrome.
When Roche pulled back on its R&D, it ended its involvement and financial support for STX209. We understand there have been issues with the therapy’s trial results, but it had statistically relevant improvements to secondary outcome measures. Luca Santarelli, head of neuroscience research at Roche has been quoted as saying “We concluded that arbaclofen wasn’t going to provide that real difference for patients.” Dr. Schwan, we respectfully disagree, the improvements we have seen in our children have changed the lives of our families.
Without Roche’s backing, on May 15, 2013, Seaside Therapeutics was forced to issue the following statement to the clinics involved in the administration of Arbaclofen:
We regret to inform you that Study 209FX303 [An Open-Label Extension Study to Evaluate the Safety, Tolerability, and Pharmacokinetics of STX209 (Arbaclofen) in Subjects with Fragile X Syndrome] is being terminated immediately. The closure of the study is due to resource limitations at Seaside Therapeutics, Inc., and is not related to any known safety issues in patients dosed with STX209.
As of July 1, 2013, none of our children will have access to STX209. The abrupt end of the study have left some of our children to suffer withdrawal symptoms. But that is hardly a consideration compared to what is about to happen to us all. We’d like to share with you the thoughts of just a few of the families in the trial.
From Cortney AbouElSeoud, mother of Ayden, age 5:
“A little background first. Ayden had a 5-word vocabulary, but seldom spoke. Within one week of treatment with Arbaclofen, his tantrums diminished noticeably. Within a month, he started using words spontaneously. Now seven months into treatment, he has surpassed development goals in every area. He speaks in small sentences, can isolate the sounds of the alphabet and can trace all his upper and lowercase letters. On Arbaclofen, he could enter first grade on par with his peers.”
“I’ve often heard fragile X described as having the sound of a vacuum in your head. Consider going through life daily with that sound and attempting to speak or to walk into a large room full of people. Imagine how difficult it would be and how easily you could become frustrated. Imagine then that there was something you could do to shut that noise off. That is how I look at Arbaclofen…. Now we have to turn that switch back on. Devastated does not even begin to describe the emotions I am feeling. Terrified. Worried. Overwhelmed…. And the tears. Because they can’t find enough money to finish the trial, I am facing a reality where I may never again hear my son say “mommy.”
From Deanna Acree, mother to Kolton Acree, age 5:
“Imagine your child is unable to tell you their tummy hurts; unable to make friends at school even though they love other kids; unable to run a quick errand with you because the store may over-stimulate them; unable to look at you at night while you tuck them into bed. Just imagine! Your child is locked in a world that no one can understand; they try and try but everyday life is a struggle. Now imagine your child is sick, he tells you what hurts; he goes to school, and has friends high fiving him in the hallway; he hugs you before bedtime and whispers ‘I love you.’ How do you look at your child and tell them their time in this world is over? How can you possibly explain that they have to go back to that frightening, isolating world of their own?”
From Aimee & Josh Freedman, parents to Nathan & Zakira Freedman, age 7 & 12:
“Parents of children with special needs are often told that their feelings of grief are similar to the feelings of a death of a loved one. You need to mourn the life that you had envisioned for your child and your family and start a whole new dream or reality. We had already said our goodbyes to the life we thought we were going to have. After 20 months of watching Nathan progress incredibly, we are now told it will all be taken away… sorry folks, no money for your child to keep taking a medication that has been absolutely life changing. Now we must mourn again, once simply wasn’t heartbreaking enough. Now that we have seen our child happy; learning to read; talking so much; having haircuts that we don’t have to hold him down for; waking up smiling instead of screaming… now that we have seen it, we will have to watch it all taken away. How can anyone watch their child regress into a world that is so hard and uncomfortable for them that all they can do is yell?”
From Tiffany Pace, mother to David & Jonathan Wright, ages 13 &6:
“I’ve seen so many amazing things in the time since David started Arbaclofen. He started reading! Not just sight words he’d memorized, but really reading! We never thought he’d learn to read at all. He started doing math problems on his own. His handwriting became legible. He began participating in class. A year ago, we were planning his very first, EVER, birthday party, with friends! He was able to tell me who he wanted to invite. For the past two years, he let us sing “happy birthday” to him – that was never possible before Arbaclofen. He hugs and kisses me… he hugs and kisses his grandparents! When he was little, I couldn’t even get him to hold my hand when crossing a street – that kind of small touch caused a complete meltdown. He even hated being held as a baby. Now he chooses to hug me! I am completely terrified at the thought of losing Arbaclofen. What if his rages come back? What if all these new skills go away? I don’t know how to explain to David what is about to happen. To either of my sons. What will happen to my children when Arbaclofen is taken away?
From Melissa Welin, mother to Caleb Welin, age 9:
“I woke up the morning after I learned that the trial for STX209 was over and stood crying at the counter while making Caleb’s breakfast – which is always accompanied by a spoon of applesauce and his morning meds – knowing that we were one dose closer to the end. I know nothing in this business is guaranteed; there’s a lot of money in pharmaceuticals because it’s expensive work. Changing the world takes lots and lots of money. I refuse to believe that this is as far as the story goes. I’ve never expected a fairy tale ending, but this defies logic. We are almost there. In the face of incredible odds, Seaside has gotten us to the point where we can see the goal line. Now we need that one person to stand up and clear the way. Please help us.”
Stop for a moment and think about what these parents consider amazing – a hug from their child; hearing the words ‘I love you;’ watching their children learn to read, make a friend, have a life. Imagine your baby is born and doesn’t want you to touch them. They grow up and can’t speak to you. They scream and yell and you can’t make them feel better. Then… there is what seems like a miracle drug. Suddenly, you are able to get to know your child for the first time. You are able to share affection and celebrate amazing milestones. Milestones parents ought to be able to take for granted.
We know that as CEO, you are responsible to your Board and to shareholders; you have to make decisions based on cost/benefit analysis and put your resources behind the most promising therapies. But please, on behalf of our children, please find a way to reinstate some funding for the study of STX209. Ideally, we’d ask that you continue the trials and explore expanded use. But at a minimum, we ask that you help Seaside provide enough supply of Arbaclofen for the patients in this study who have benefited so dramatically.
We are sharing our stories as widely as possible in the hope of finding a way of turning this nightmare into a dream-come-true. On May 30, 2013, The Boston Globe published a story and on June 6, 2013, the New York Times did as well. We have also reached out to our supporters in Congress. But we realize, ultimately, it will likely be your decision.
Please help us keep the children we have loved for so long, but only just recently had the chance to meet. Please, read the stories of the many families who are crying out for your help at Fragile X and Autism Families for STX209. We appreciate all the support and guidance you can provide.
We look forward to hearing from you with hope in our hearts.
Very sincerely,
If you would like to sign on to the letter and support our families, please enter your information below and click Submit.
Earleville, MD
Mifflinburg, PA
Ft myers, FL
Grover Beacj, CA
Mumbai, India
Westfir,
Leduc, Canada
Londrina, Brazil
Springdale, AR
Suwanee, GA
Sunnyvale, CA
sunnyvale, CA
Lawrence, KS
Oakville, Canada
London, United Kingdom
odense, Denmark
Yuba city, CA
Girne, Cyprus
New Hyde Park, NY
Cedar Rapids, IA
dianaandreescudia@yahoo.com, Guinea-Bissau
New Delhi, India
Strasburg, CO
Strasburg, CO
Redondo Beach, CA
Iselin, NJ
Englewood, CO
Denver, CO
Englewood, CO
Newark, DE
oxford, MA
Uxbridge, MA
Duldey, MA
Oakhurst, NJ
Tampa, FL
Albuquerque, NM
Etna, OH
Columbus, GA
Catalan, GA
Midland, GA
Box Springs, GA
Lakeland, FL
Ellerslie, GA
rockville, MD
Breinigsville, PA
Sutton surrey, United Kingdom
Sutton surrey, United Kingdom
Shrewsbury, VT
new london, OH
Chester, NJ
Denia, Spain
Dallas,
Zephyrhills, FL
Highland park, IL
Burlington, NJ
Norwich, CT
Cupertino, CA
Marion, NY
Kingsland, GA
Glencoe, IL
Concord, NC
Cobourg, Canada
Newnan, GA
weymouth, MA
Barnegat, NJ
Pinehurst, NC
Charlotte, NC
Charlotte, NC
columbus, OH
kolding, Denmark
MIAMI, FL
Miami, FL
Key Largo, FL
Miami, FL
buena park,
Northbrook, IL
Lewis Center, OH
Piscataway, NJ
Midvale, UT
Skokie, IL
Skokie,
Denham Springs, LA
Skokie, IL
Louisville, KY
beachwood, NJ
Milwaukee, WI
Pittston, PA
Skokie, IL
Oslo, Norway
Exton, PA
Morristown, NJ
Hønefoss, Norway
Frederiksberg, Denmark
Evanston, IL
Bjerringbro, Denmark
Copenhagen, Denmark
Bergen, Norway
Oslo, Norway
asbury, NJ
Bayonne, NJ
Titusville, NJ
Titusille, NJ
Oslo, Norway
Copenhagen, Denmark
Græsted, Denmark
Aldbury, United Kingdom
San Diego, CA
La Mesa,
Herzliya Pituach, Israel
san diego, CA
Tyne and Wear, United Kingdom
zephyr cove, NV
Templeton, CA
West Lafayette, IN
Manasquan, NJ
Denia, Spain
Spain,
Visalia, CA
Rolla, MO
Watford, United Kingdom
Visalia, CA
Denia, Spain
Rahway, NJ
San Francisco, CA
San Francisco, CA
Aurora, IL
Morgantown, WV
Chicago, IL
Visalia, CA
Visalia, CA
Lake Bluff, IL
Chicago, IL
Visalia, CA
visalia, CA
chicago, IL
Vail, AZ
Highland Park, IL
Deerfield, IL
Zionsville, IN
Oceanside, CA
Edina, MN
San Diego, CA
Las Vegas, NV
Gilbert, AZ
Tempe, AZ
Tempe, AZ
Visalia, CA
Santaquin, UT
Santaquin, UT
Tempe, AZ
vacaville, CA
vacaville, CA
vacaville, CA
vacaville, CA
Dixon, CA
vacaville, CA
Dixon, CA
Visalia, CA
Hoboken, NJ
Avondale, AZ
Avondale, AZ
Highland Beach, FL
Glendale, AZ
reading, PA
Visalia, CA
Gardnerville, NV
Watsontown, PA
Grove City, OH
Columbus, MS
Keller, TX
Salem, VA
lincolnwood, IL
Roselle, NJ
Miami, FL
Miami, FL
Springfield, MA
Sammamish, WA
Miami, FL
Princeville, IL
to, IL
Denison, KS
Middletown, IL
Seattle, WA
Alverca, Portugal
Atlanta, GA
Sidney, NY
Sahuarita, AZ
Seattle, WA
Carmel, IN
Lancaster, PA
Kenilworth, NJ
Muncie, IN
Tampa, FL
Visalia, CA
Beverly,
New Ulm, MN
Holton, KS
cincinnati, OH
Miami, FL
Pendleton, IN
Sanford, FL
Key Largo, FL
Barnegat, NJ
Manahawkin, NJ
Dakota Dunes, SD
Lexington, IL
Kannapolis, NC
Natick, MA
Visalia, CA
Prairieville, LA
Indianapolis, IN
Greensburg, PA
Holton, KS
Woking, United Kingdom
Aldbury, United Kingdom
Visalia, CA
Holton, KS
Airdrie, Canada
Holton, KS
Wolcott, IN
Richardson, TX
Airdrie, Canada
Overland Park, KS
Greensburg, PA
Latrobe, PA
Holton, KS
Holton, KS
Jeannette, PA
Madison Heights, VA
Visalia, CA
barnegat, NJ
Essex Ontario, Canada
Mount Pleasant, PA
iselin, NJ
Holton, KS
mayetta, KS
Fieldon, IL
Kannapolis, NC
Holton, KS
Concord, NC
portland, SC
Holton, KS
Lacombe, Canada
Baton Rouge, LA
Essex, Canada
Liberty Township, OH
Baton Rouge, LA
Flat Rock, MI
Cincinnati, OH
Oxford, OH
Yorkville, IL
McDonough, GA
Phila, PA
Essex, Canada
Cincinnati, OH
Barnegat, NJ
SEVEN MILE, OH
Middletown, OH
Brighton, MI
Seabrook, NH
Houston, TX
Northfield,
Elwood, IN
Cincinnati, OH
Uniontown, OH
LITTLE SILVER, NJ
Fishers, IN
Forked River, NJ
Flagstaff, AZ
Noblesville, IN
carmel, IN
Edison, NJ
chicago, IL
williamsburg, MI
Hayle, Cornwall, United Kingdom
Grandville, MI
Olathe, KS
Powell, TN
Columbia, SC
Dorset, United Kingdom
Holton, KS
Kansas City, MO
barnegat, NJ
London, United Kingdom
Manchester, United Kingdom
wilmington, NC
Davenport, FL
Forked River, NJ
Baltimore, MD
Le Roy, IL
Le Roy, IL
moultonboro, NH
barnegat, NJ
Philadelphia, PA
little egg harbor, NJ
Muncie, IN
Le Roy, IL
Lake Forest Park, WA
Eastland, TX
Easton, PA